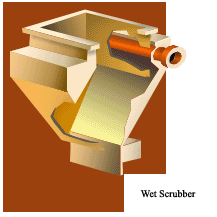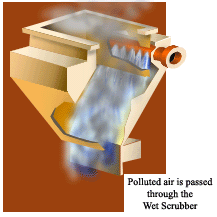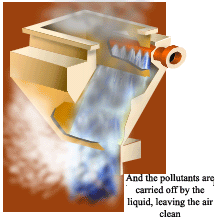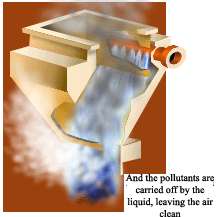
 Scrubbers or wet collectors remove particles or gases from the exhaust stream by using water sprays. Gases can be absorbed if they are water-soluble or by adding various chemicals to the spray. Particles of dust or soot can also be captured in microscopic liquid mists. Before the exhaust leaves the scrubber, the liquid mists must be collected before the exhaust enters the public air.
Scrubbers or wet collectors remove particles or gases from the exhaust stream by using water sprays. Gases can be absorbed if they are water-soluble or by adding various chemicals to the spray. Particles of dust or soot can also be captured in microscopic liquid mists. Before the exhaust leaves the scrubber, the liquid mists must be collected before the exhaust enters the public air.
Generally, high-powered scrubbers remove more particles but are more costly to operate due to added energy costs. Scrubbers that remove gases like sulfur oxides, nitrogen oxides or hydrochloric acid depend more heavily on the mechanical and chemical engineering design, not as much on power. Scrubbers are generally better at removing particles than cyclones, but not as good as electrostatic precipitators or baghouses unless operated at high power.
Scrubbers collect microscopic particles by injecting fine mists of water into the exhaust. The moving particles cannot avoid impacting into the droplets, making them easier to collect. Scrubbers must do two things: bring pollutants into contact with the water mist, then remove the airborne water mist. Removing mist sounds simple, but the water droplets are extremely small and the air flows through the scrubber very quickly.
The DNR air quality engineers use physics, chemistry and engineering skills to determine how the system will operate before an air quality permit is issued. The DNR also works with professionals from the business too. Temperature, flue gas and pollutant composition, air pressure, solubility and the chemical reactivity must be calculated to figure out how much pollution will be removed.
Scrubbers come in all shapes and sizes. Sometimes chambers are filled with water spray nozzles. Others use complex systems of baffles, motors, sprays and nozzles. Others use chambers packed with small, odd shaped fill material to increase surface area for particles and water to hit for collection. Some force pollutants through a small nozzle passage to increase the gas flow as it squeezes through the small opening. Here water spray is injected and sheared into fine droplets for the particles to hit.
Scrubbers offer many design options to meet a variety of air pollution control needs. For example, "flue gas desulfurization" scrubbers inject lime or limestone to react with sulfur dioxide gas to form sulfates, which are then removed.
Scrubber benefits:
Can collect both particles and gases and can handle high temperature gases. Fire and explosion hazards found in some dry-collection systems are eliminated with wet collection. Once the pollutants are collected, they cannot escape easily, unlike dry collection systems where dust can be released from hoppers. The water slurry can sometimes be easier to handle than dry dust.
Scrubbers also have disadvantages:
Water and absorbed gases can become very corrosive, so the scrubber must be properly designed to meet each specific industrial process. Because scrubbers use water, high-humidity air leaving the scrubber can cause large water vapor plumes when emitted into cold Iowa winter air. Fog and precipitation can cause local meteorological problems or driving hazards near the industry. And because water is used to clean the air, the dirty water also needs to be cleaned. Settling ponds and sludge handlers are often needed to clarify the water slurry. High-powered scrubbers are costly to operate when using high fan speeds.